 Jefferson County Public Health (JCPH), in collaboration with the Colorado Department of Public Health and Environment (CDPHE), is advising the public about a potential exposure to hepatitis A at the King Soopers located at 5301 W. 38th Avenue in Wheat Ridge, Colo. A food handler at this location has tested positive for hepatitis A, a
Jefferson County Public Health (JCPH), in collaboration with the Colorado Department of Public Health and Environment (CDPHE), is advising the public about a potential exposure to hepatitis A at the King Soopers located at 5301 W. 38th Avenue in Wheat Ridge, Colo. A food handler at this location has tested positive for hepatitis A, a
Food Poisoning Watch
Public Health Alerts Residents of Possible Exposure to Hepatitis A at Sunlife Organics Juice Bar in West Hollywood California
 Come on CDC, let’s call for hepatitis A vaccines for all food service workers.
Come on CDC, let’s call for hepatitis A vaccines for all food service workers.
There has been an ongoing and massive hepatitis A outbreak that has been sweeping the United States over the last several years. Seeing yet another hepatitis A scare in a food service worker has been a far too common occurrence. Here…
Ready-to-Eat (RTE) chicken salad recalled due to plastic pieces
 The U.S. Department of Agriculture’s Food Safety and Inspection Service (FSIS) is issuing a public health alert for a ready-to-eat (RTE) chicken salad product containing a Food and Drug Administration (FDA) regulated salad dressing that has been recalled by the producer due to concerns that the salad dressing may contain hard plastic. FSIS is issuing…
The U.S. Department of Agriculture’s Food Safety and Inspection Service (FSIS) is issuing a public health alert for a ready-to-eat (RTE) chicken salad product containing a Food and Drug Administration (FDA) regulated salad dressing that has been recalled by the producer due to concerns that the salad dressing may contain hard plastic. FSIS is issuing…
Hepatitis A scare at Saskatchewan Tim Hortons
 The Saskatchewan Health Authority (SHA) is alerting the public to a risk of exposure to Hepatitis A from a food service employee at a restaurant located in Emerald Park, SK.
The Saskatchewan Health Authority (SHA) is alerting the public to a risk of exposure to Hepatitis A from a food service employee at a restaurant located in Emerald Park, SK.
The SHA (Regina area) is conducting an investigation and follow-up of a case of Hepatitis A in a food handler who assisted with all meal…
Hepatitis A risk at KFC in Hudson North Carolina
 The State of North Carolina notified Caldwell County this late afternoon about a confirmed case of Hepatitis A in an individual who works at KFC in Hudson.
The State of North Carolina notified Caldwell County this late afternoon about a confirmed case of Hepatitis A in an individual who works at KFC in Hudson.
According to state guidelines, transmission to patrons is unlikely, so vaccination for restaurant patrons is not recommended at this time.
“We are working closely with the restaurant, the…
FDA: Salmonella outbreaks linked to Onions and Seafood and an E. coli outbreak linked to Spinach ongoing – A new Salmonella Outbreak brewing

4,000 exposed to Hepatitis A positive Starbucks employee in New Jersey
 In light of the case of hepatitis A in an employee at a local Starbucks, the Camden County Health Department will be administering vaccines to anyone who feels they may have been exposed at the Camden County Health Hub located at 200 College Drive in Blackwood.
In light of the case of hepatitis A in an employee at a local Starbucks, the Camden County Health Department will be administering vaccines to anyone who feels they may have been exposed at the Camden County Health Hub located at 200 College Drive in Blackwood.
Today’s clinic will operate from 10 a.m. to 2…
Backyard Chickens hospitalize 273 and kill 2 with Salmonella – 1,135 sick in 2021 including 268 under the age of 5
 CDC: This year’s investigation of outbreaks linked to backyard poultry is over. However, any backyard poultry can carry Salmonella germs that can make you sick. Always take steps to stay healthy around your flock.
CDC: This year’s investigation of outbreaks linked to backyard poultry is over. However, any backyard poultry can carry Salmonella germs that can make you sick. Always take steps to stay healthy around your flock.
CDC and public health officials in several states investigated multistate outbreaks of Salmonella infections with serotypes of Enteritidis, Hadar, Indiana, Infantis,…
Hey Starbucks, you should pay the cost of the hepatitis vaccines and time loss standing in line due to your ill employee
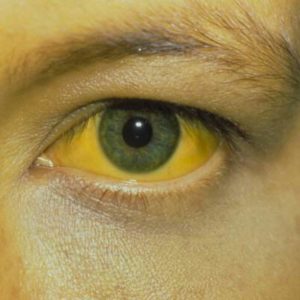 I got my eye on you!
I got my eye on you!
Jasmine Payoute of Philadelphia CBS reports that there are growing worries after a Starbucks employee tested positive for hepatitis A in Gloucester Township. There’s now a vaccination clinic provided by Camden County for anyone who feels they may have been exposed.
The line wrapped around the corner Friday afternoon…
Hey Starbucks, vaccinate your employees for Hepatitis A so they don’t risk exposing your customers
 The Camden County Health Department has been notified by a health care provider that a food handler employed at a Starbucks at 1490 Blackwood Clementon Road in Gloucester Township tested positive for hepatitis A and worked through the infectious period.
The Camden County Health Department has been notified by a health care provider that a food handler employed at a Starbucks at 1490 Blackwood Clementon Road in Gloucester Township tested positive for hepatitis A and worked through the infectious period.
On Wednesday, Nov. 17, the Department of Health was notified that a patient tested positive…